Innokas and Vitalthings: Synergistic collaboration in class IIb contactless health monitor development
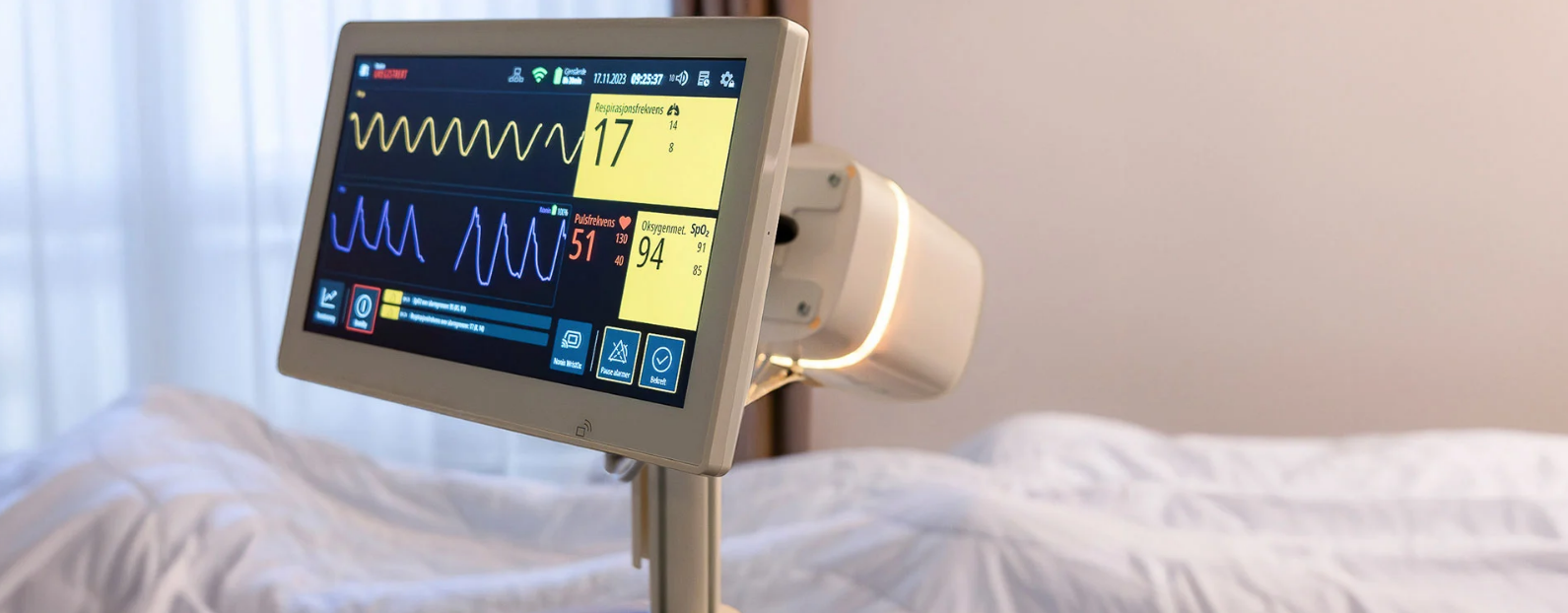
Developing complex medical devices requires adaptability, expertise, a deep understanding of regulatory requirements, and collaboration. That’s exactly what Innokas brought when partnering with Vitalthings, a Norwegian HealthTech company specializing in contactless health monitoring. Together, they developed Guardian M10, an innovative respiratory monitoring device for hospitals and healthcare institutions, now officially MDR-certified as a Class IIb medical device.
Vitalthings selected Innokas as a development partner to help navigate the technological, regulatory, and time-to-market challenges of bringing Guardian M10 to life. The collaboration began in early 2021 with an initial pre-study phase, where Innokas supported concept development, regulatory planning, and technical feasibility assessments. As the project evolved, Innokas expanded its involvement, guiding the product specification, productization, and eventual transition to manufacturing. Close coordination between Innokas and Vitalthings was essential in overcoming challenges and keeping development aligned with market needs.
The successful partnership resulted in a fully certified, market-ready product. With Guardian M10 now in production, Innokas and Vitalthings continue their cooperation on future innovations.
Source: Innokas Medical
